Outputs
D6.1 - Website Twitter and LinkedIn accounts live
17/10/2025Public deliverable.
The document describes the motivation behind the concept of the website and the objectives as a key tool for dissemination and communication actions. The document provides a description of the public site, defining also the social media tools used, such us Twitter and LinkedIn.
Read the deliverable here.
D4.2 - Network-wide training events report - R1
17/10/2025Public deliverable
The document describes the activities carried out during the first D-Carbonize Workshop & kick-off meeting co-organized by ICIQ and RUG (24-25 January, Tarragona, Spain).
Read the deliverable here.
D4.3 - Network-wide training events reports - R2
17/10/2025Public deliverable.
The document describes the activities carried out during the second D-Carbonize Workshop organized by UISCR-CNRS (14-15 October, Rennes, France).
Read the deliverable here.
Solvent-Free Synthesis of Renewable FDCA-based Bis-Cyclic Carbonate Using a Metal-Free Heterogeneous Catalyst
19/02/2026Giovanni Berluti - DC6
In this work, we developed a new, fully renewable catalytic strategy to synthesise an industrially relevant bio-based cyclic carbonate: 2,5-bis-dicarboxyl furan cyclic carbonate (BDFCC). This compound contains two terminal cyclic carbonate groups, which makes it attractive for application as a building block for green polymers. BDFCC was prepared from two renewable compounds: the methyl ester of 2,5-furan dicarboxylic acid and glycerol carbonate, via a transesterification reaction at mild temperature (80 °C) using a novel, metal-free dual catalytic system consisting of commercially available ion-exchange resin beads (Amberlite IRA900-Cl) and glycidol. Under conditions previously reported for homogeneous catalysis, involving nhexane as solvent, only a moderate yield of 43% was achieved. A major improvement in both performance and sustainability of the process was achieved by implementing an equilibrium-shifting solvent-free strategy, which overcomes the thermodynamic limit of the reaction (ΔG 0 = +25 kJ mol -1 based on DFT calculations). This approach, combined with an optimisation of the reaction conditions, allowed increasing the BDFCC yield to 86%. Furthermore, the heterogenous resin bead catalyst was recyclable over multiple runs without experiencing loss of activity. Control experiments allowed to gain insight in the reaction mechanism and revealed the crucial role played by glycidol in promoting the catalytic activity of the Amberlite resin beads.
Click here to read the article.
CO2/epoxides ring-opening copolymerization towards hydroxy-functionalized polycarbonates
04/02/2026Nishant Chaudhary - DC8
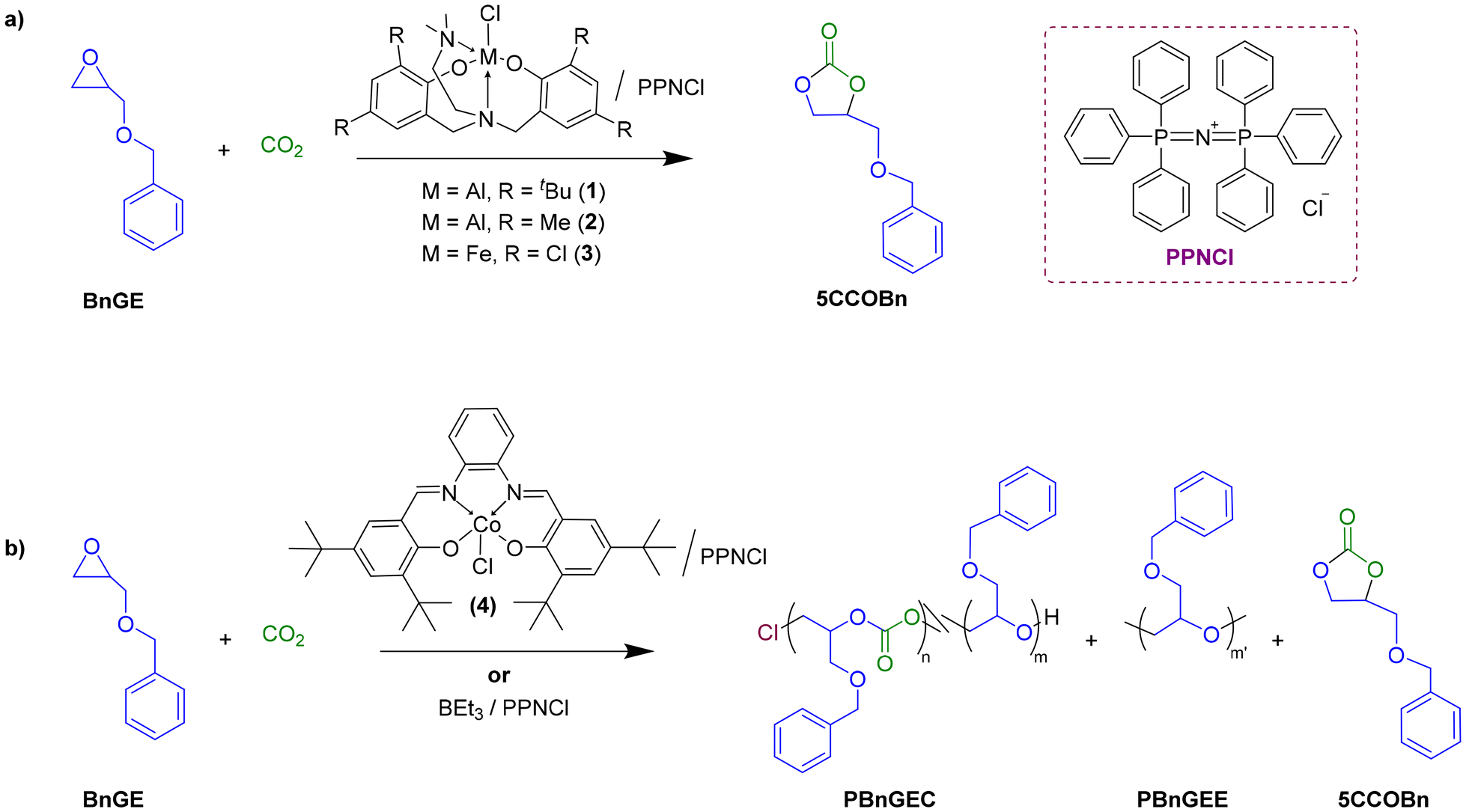
Ring-opening copolymerization (ROCOP) of CO2 and epoxides witnesses continued interest to access sustainable polycarbonates. Introduction of an exocyclic functional group onto the epoxides enables to tune and diversify the properties of the resulting CO2-based polycarbonates. Herein, the CO2/benzyl glycidyl ether (BnGE) or CO2/cyclohexene oxide (CHO) ROCOP has been performed, using a bicomponent catalyst system composed of either a {diamino-bisphenolate}MCl (Al, Fe) or {Salphen}CoCl complex or triethylborane (BEt3) as catalyst, combined with bis(triphenylphosphoranylidene)ammonium chloride (PPNCl) as initiator. While the Al/Fe-based catalyst systems selectively returned the corresponding benzyloxymethylene five-membered cyclic carbonate (5CCOBn) with poor activity in the copolymerization of CO2/BnGE, the {Salphen}CoCl/PPNCl and BEt3/PPNCl systems produced poly(benzyl glycidyl ether carbonate) (PBnGEC) with high chemoselectivity (∼80% and >98%) and regioselectivity (>99% and ∼84%), featuring >99% and ∼85% of carbonate linkages, respectively. Investigation of the {Salphen}CoCl/PPNCl and BEt3/PPNCl catalytic systems in the ROCOP of CO2/BnGE/CHO with different comonomers loadings, enabled to prepare a series of tunable P(BnGEC-co-CHC) terpolymers with similar selectivities. Subsequent hydrogenolysis of these hydrophobic polymers using Pd/C resulted in the deprotection of the side-chain benzyloxy moieties, affording the corresponding hydrophilic P(GC-co-CHC) polymers featuring hydroxyl pendant groups; yet, significant degradation of the polycarbonate main chain was observed for hydroxyl contents >15 mol%. Depolymerization of PBnGEC, PCHC and P(BnGEC-co-CHC) using 1,5,7-triazabicyclo[4.4.0]dec-5-ene (TBD) as catalyst quantitatively returned both corresponding 5CCs. In-depth characterizations by NMR spectroscopy, mass spectrometry, SEC, TGA, and DSC analyses supported well-defined protected and deprotected terpolymers with tunable chemical and thermal properties, providing opportunities for biomedical and/or industrial outcomes.
Click here to read the article.
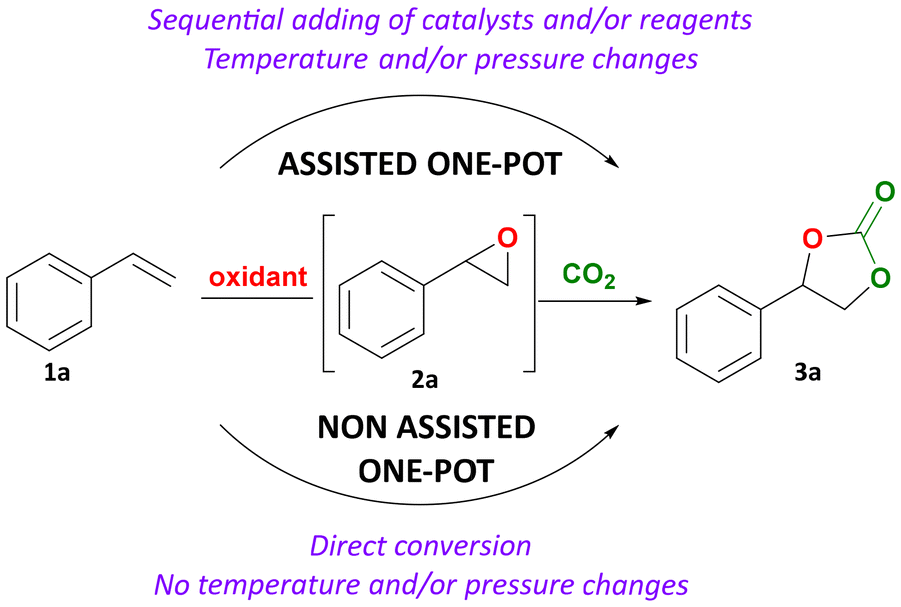
A one-pot organocatalytic process for the synthesis of cyclic carbonates from CO2 and alkenes using cumene hydroperoxide as a green oxidant
03/03/2026Angelo Scopano - DC5
Cyclic carbonates are usually obtained from coupling of carbon dioxide and epoxides. The latter are generally prepared through the selective oxidation of alkenes or other compounds containing a double bond. However, a one-pot route in which an alkene is directly converted into a cyclic carbonate would be preferable as it would circumvent the handling of generally toxic epoxides and would increase process efficiency in terms of energy, solvent and reagents usage. Here, we present an attractive strategy combining a recyclable oxidant (cumene hydroperoxide, CHP) with an inexpensive, metal-free organic halide salt as catalyst. These components act cooperatively promoting the oxidation of the chosen model substrate (styrene) and the cycloaddition of CO2 to the generated epoxide intermediate. Tetrabutylammonium bromide exhibited the best catalytic performance, providing a 55% styrene carbonate yield after 6 h at 10 barg of CO2 and 80 °C using 1.5 equivalents of oxidant; and 67% in the presence of 4 equivalents of oxidant. These cyclic carbonate yields are significantly higher than those obtained with other oxidants (tert-butyl hydroperoxide and hydrogen peroxide). A scope of substrates was converted into their respective cyclic carbonates including a new bio-based methylisoeugenol-derived product and a cyclic carbonate attained from bio-based methyl oleate (having a disubstituted double bond). From mechanistic control experiments, we determined that the oxidation step proceeds through a radical mechanism, with an active involvement of CHP in epoxide activation via hydrogen-bonding, demonstrating a dual role of the oxidant. Our strategy offers a practical proof of concept of a direct approach to cyclic carbonates with a simple organocatalyst that could be reused in four consecutive runs with a similar performance, and using a recyclable oxidant.
Click here to read the article.
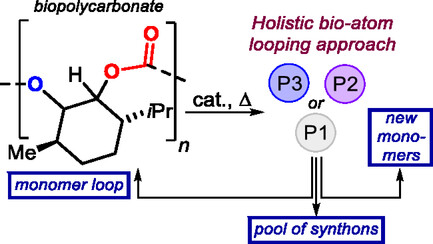
Enabling a Diversity-Oriented Catalytic Atom Looping of a Biobased Polycarbonate
06/10/2025Click here to read the article.
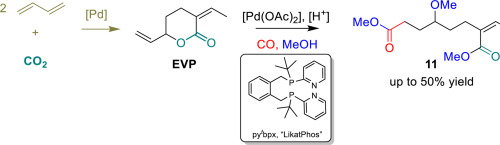
Palladium-catalyzed methoxycarbonylation of a commodity CO2-sourced δ-valerolactone
07/10/2025Domino Synthesis of Functionalized Cyclic Acetals From Organic Carbonates
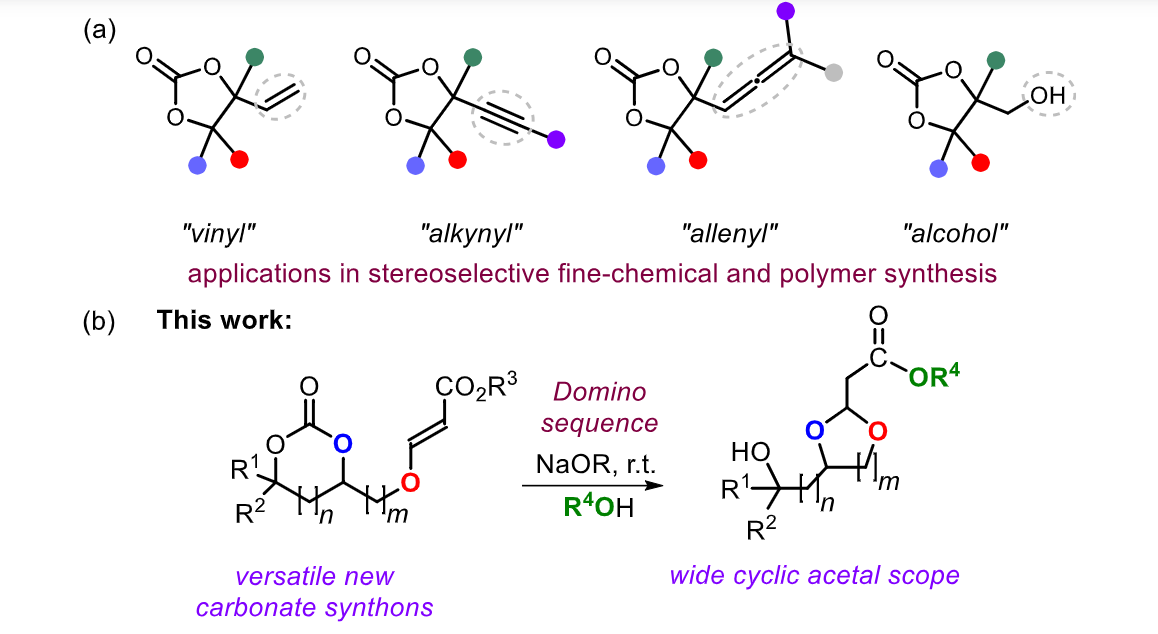
04/12/2025Natalia Kulbacka - DC2
We report a method for the base-mediated transformation of ether-tethered acrylic ester-based cyclic carbonates into functionalized cyclic acetals. The protocol builds on the use of hydroxyalkyl-substituted cyclic carbonates that undergo an oxa-Michael addition reaction in the presence of alkyl propiolates thereby forging (E)-configured acrylic ether intermediates. The scope of the reaction involves the use of both five- and six-membered cyclic carbonates, and correspondingly, both five- andsix-membered cyclic acetals can be prepared. The amount of reagents, the purification method, and the type of ester substrate all contribute to the efficiency of the transformation. Mechanistic control reactions point at the intermediacy of an alkoxide that induces an intramolecular Michael addition onto the acrylic double bond following alkoxide-mediated formation of both an alcohol and ester in the final product. These functional groups, among others, further enable easy diversification of acetal-based synthons.
Click here to read the article.
.png)
_1760689135.png)
_1760689697.png)